
CDC Narrows Vaccine Recommendations In Response To Trump Order
Authored by Zachary Stieber via The Epoch Times,
Health officials announced on Jan. 5 that they’re narrowing the number of vaccines recommended broadly for children in response to a recent order from President Donald Trump.
The Centers for Disease Control and Prevention (CDC) is moving forward with only broad recommendations for eight vaccines for children, down from 14.
Trump, in December 2025, directed Health Secretary Robert F. Kennedy Jr. and acting CDC Director Jim O’Neill to review vaccine schedules in the United States and peer countries and determine if the U.S. schedule should be updated.
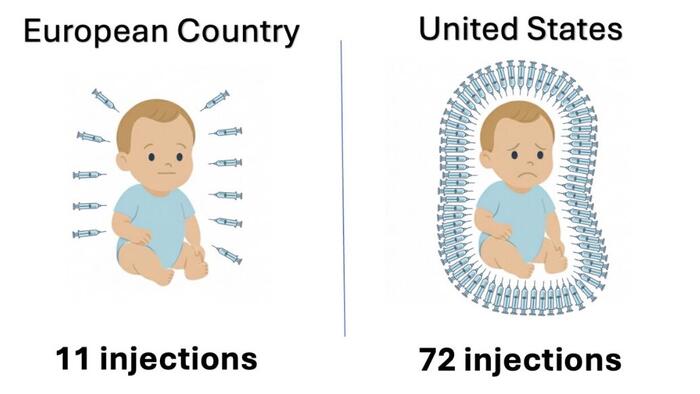
He named three countries, including Denmark, that recommend fewer vaccines and fewer vaccine doses.
“President Trump directed us to examine how other developed nations protect their children and to take action if they are doing better,” Kennedy said in a statement.
“After an exhaustive review of the evidence, we are aligning the U.S. childhood vaccine schedule with international consensus while strengthening transparency and informed consent. This decision protects children, respects families, and rebuilds trust in public health.”
Moving forward, the CDC will stop broadly recommending vaccines against influenza, rotavirus, hepatitis A, and meningococcal disease. The CDC in 2025 already narrowed recommendations for hepatitis B and COVID-19 vaccination based on advice from advisers selected by Kennedy. The agency is maintaining its recommendation that children whose mothers did not receive a respiratory syncytial virus vaccine receive an antibody, or passive immunization, against the virus.
The old schedule can be viewed here, and the new schedule can be viewed here.
The changes were recommended by Dr. Tracy Beth Hoeg, acting director of the Food and Drug Administration’s (FDA’s) Center for Drug Evaluation and Research, who, during a recent presentation, commented favorably on Denmark’s vaccine schedule, and Martin Kulldorff, whom Kennedy appointed a senior adviser in 2025. Hoeg and Kulldorff said in a 34-page assessment that an update was needed because of falling trust in public health, decreases in vaccination rates, and evidence that some recommended vaccines had limited benefits.
A CDC official told reporters on a call on Jan. 5 that the agency consulted with officials in Denmark, Germany, and Japan, as well as vaccine scientists at the CDC and FDA.
Vaccine manufacturers were not consulted, another official said.
The administration says the update does not prevent children from accessing vaccines and that insurers will continue to cover them without cost-sharing under the Affordable Care Act.
The CDC still recommends some of those vaccines for certain populations, such as hepatitis B vaccination for children born to women who test positive for the virus. For others, it is focused on shared clinical decision-making or recommending that people consult doctors and consider factors such as the risk of illness when deciding whether to have their children vaccinated.
The CDC is keeping in place broad recommendations for vaccines against diphtheria; tetanus; acellular pertussis, or whooping cough; haemophilus influenzae type b; pneumococcal disease; polio; measles; mumps; rubella; varicella, also known as chickenpox; and human papillomavirus (HPV).
The new schedule lowers the number of recommended HPV doses from two to one, after some recent research indicated that one dose is as effective.
“Important vaccines … will be continued to be recommended for our children,” a Department of Health and Human Services official said on the call.
“This change is going to spark major pushback, but that reaction was inevitable,” Dr. Joel Warsh, a pediatrician based in California, told The Epoch Times in an email.
“Reducing universal recommendations doesn’t mean vaccines are being banned or declared unsafe—it means the CDC is finally acknowledging that not every vaccine has the same risk-benefit profile for every child.”
The American Academy of Pediatrics said it opposed the changes.
“At a time when parents, pediatricians, and the public are looking for clear guidance and accurate information, this ill-considered decision will sow further chaos and confusion and erode confidence in immunizations,” Dr. Andrew Racine, president of the group, which partners with vaccine companies, said in a statement.
“This is no way to make our country healthier.”
President Trump took his social media account to explain…
Tyler Durden
Mon, 01/05/2026 – 18:25

